Welcome to your GRADE 12 CHEMISTRY MODEL EXAMINATIONS 2017
CHEMISTRY MODEL EXAMINATION FOR GRADE 12
JAN 2017
PHYSICAL CONSTANTS
a)Gas constant, R=8.314J/molK = 0.0821L.atm/molK
b)Avogadro’s number = 6.02x1023mol-1
c)Planck’s constant, h = 6.63x10-34Js
d)Speed of light, C=3x108m/s
e)Faraday’s constant(F)=96500C/mol
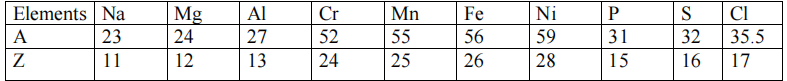
ATOMIC NUMBERS(Z) AND ATOMIC WEIGHTS(A)
I.CHOOSE THE CORRECT ANSWER FROM THE GIVEN ALTERNATIVE(S)
1.
According to the Brønsted-Lowry theory, a base is defined as:
2.
Which of the following solid is Not amorphous solid?
3.
In an ionic bond, electrons are:
4.
What will happen to the equilibrium position if OH- ions are added to a solution of acetic acid (CH₃COOH ⇌ CH₃COO⁻ + H⁺)?
5.
Which of the following is true about the liquid state of matter?
6.
What is true for the following given reaction?
Cr₂O₇²⁻ + 14H⁺ + 6Cl⁻ → 2Cr³⁺ + 6Cl₂ + 7H₂O
7.
What is the conjugate base of H₂CO₃?
8.
Which gas law relates the pressure and volume of a gas at constant temperature?
9.
Which of the following best describes metallic bonding?
10.
Given a 0.1 M solution of a weak acid (Ka = 1.8 × 10⁻⁵), what is the approximate pH of the solution?
11.
What happens to the kinetic energy of particles as a substance moves from a solid to a liquid state?
12.
What type of intermolecular force is responsible for the high boiling point of water?
13.
Why does a buffer solution resist changes in pH? Because
14.
What is the wavelength of the yellow sodium emission, which has a frequency of 5.09 × 10¹⁴ s⁻¹?
15.
In a covalent bond, how do atoms achieve a stable electron configuration?
16.
Which of the following is true about the strength of metallic bonds?
17.
Which of the following factors affects the ionization of weak acids?
18.
The blue color in fireworks is often achieved by heating copper(I) chloride to about 1200°C. Then the compound emits blue light having a wavelength of 450nm. What is the increment of energy (the quantum) that is emitted at 4.50 × 10² nm by CuCl?
19.
In a molecule of nitrogen (N₂), how many electrons are shared between the two nitrogen atoms?
20.
If the reaction, Cu + HNO₃ → Cu(NO₃)₂ + NO₂ + H₂O is balanced, what is the coefficient of HNO₃?
21.
A sample of gas occupies 10.0 L at 2.0 atm pressure. What will be its volume if the pressure is reduced to 1.0 atm at constant temperature?
22.
A salt derived from a weak acid and a weak base will produce a solution whose pH depends on:
23.
At STP, 2 moles of an ideal gas will occupy:
24.
A student electrolyzes an aqueous copper sulfate solution using platinum electrodes. What will they observe at the anode?
25.
If a titration curve shows a sharp pH rise around pH 9, what type of titration is it?
26.
Which of the following is the correct order of elements in the Modern Periodic Table?
27.
Which law states that mass is neither created nor destroyed in a chemical reaction?
28.
Which of the following is a key characteristic of chemical industries?
29.
What is a major environmental concern related to cement manufacturing?
30.
According to Dalton's atomic theory, atoms of the same element are:
31.
What is the main raw material obtained from the atmosphere for industrial use?
32.
One of the advantages of periodic classification is that it helps in understanding:
33.
Which subatomic particle has a mass closest to that of a proton?
34.
What role does industrial chemistry play in society?
35.
Which of the following elements is correctly matched with its block belongs to in the periodic table?
36.
What does the Aufbau principle state about the filling of electron orbitals?
37.
Which natural resource is most crucial for energy production in developing countries?
38.
Which industry significantly contributes to Ethiopia's agricultural economy?
39.
Which of the following best describes the Rutherford gold foil experiment?
40.
What is the primary industrial process for manufacturing ammonia?
41.
Why does the electron configuration of chromium (Cr, atomic number 24) deviate from the expected [Ar] 4s² 3d⁴?
42.
Which renewable resource can directly be used for generating electricity?
43.
Which of the following is the correct value of the angular momentum quantum number (l) for a d-orbital?
44.
Which resource is classified as non-renewable?
45.
How many unpaired electrons are present in the electron configuration of carbon (1s² 2s² 2p²)?
46.
Which raw material is primarily used in glass manufacturing?
47.
Which of the following forms of electromagnetic radiation has the shortest wavelength?
48.
Which local Ethiopian alcoholic beverage is commonly made using traditional methods?
49.
When an electron in a hydrogen atom jumps from the n = 3 level to the n = 2 level, what happens to the energy of the photon emitted?
50.
What is used in the Contact Process for sulfuric acid production?
51.
Which method is commonly used to preserve food in Ethiopia?
52.
What is the main purpose of tanning in leather production?
53.
What byproduct of sugar manufacturing is used to produce ethanol in Ethiopia?
54.
Which material is added to cement to control its setting time?
55.
What does the period number of an element indicate in the Modern Periodic Table?
56.
What is the primary product of sugar manufacturing?
57.
Which of the following is NOT a characteristic of transition elements?
58.
Which glass type is commonly used in laboratory equipment due to its resistance to chemical corrosion?
59.
Which ions are preferentially discharged at the cathode during the electrolysis of copper (II) sulfate using platinum electrodes?
60.
Which type of crystal structure would be best suited for a substance that is hard, has a high melting point, and is a good insulator?
61.
Why are isotopes of an element chemically similar?
62.
A student is asked to balance the redox reaction using the half-reaction method. What is the first step?
63.
Which of the following is the strongest type of chemical bond?
64.
Who discovered the nucleus of an atom?
65.
What determines the ions preferentially discharged during electrolysis?
66.
According to Bohr's atomic model, where are electrons located?
67.
What happens if the salt bridge in a voltaic cell is removed?
68.
Design an experiment to determine the mass of silver deposited when a current of 2 A is passed through silver nitrate for 30 minutes. What formula would you use?
69.
Why do ionic compounds have high melting points?
70.
Who proposed the first atomic theory based on experimental evidence?
71.
Which of the following is NOT a common industrial application of electrolysis?
72.
Which of the following molecules would likely exhibit sp³ hybridization in its central atom?
73.
Which model of the atom is referred to as the "plum pudding model"?
74.
Which bonding theory explains the formation of covalent bonds through the overlap of atomic orbitals?
75.
Which of the following molecules has a linear molecular geometry?
76.
Create a cell diagram for a voltaic cell using Zn and Cu electrodes in their respective sulfate solutions. How will it be represented?
77.
Which type of covalent bond is formed when electrons are shared equally between two atoms?
78.
What is the hybridization of the central atom in a molecule of carbon dioxide (CO₂)?
79.
A current of 5A flows for 2 hours during electrolysis. What is the total charge passed?
80.
What is the bond angle in a molecule with a tetrahedral geometry, such as methane (CH₄)?