Welcome to your GRADE 12 CHEMISTRY MODEL EXAMINATIONS 2015
GRADE 12 CHEMISTRY MODEL EXAMINATIONS
MESEKEREM 2015/ SEPTEMBER 2022
DIRECTIONS: Each of the following questions is followed by four possible alternatives. Read each question and carefully blacken the letter of your best choice on the separate answer sheet provided.
1.
The reaction of an anion or a cation of a salt, or both, with water is known as______.
2.
During the laboratory session, the 12th grade student named Etsube made an error while conducting an experiment. In which part of the laboratory report writing did she indicate such an error?
3.
Which pairs of color and wave contains highest wave length?
4.
Which of the following is true about CO₂ molecule?
5.
Which of the following is correct about the name Nylon 66 co-polymer?
6.
The standard state of Aluminum, Nitrogen and Water are ______ respectively.
7.
Which batteries are faced with leak out and environmental problem respectively?
8.
Which substance is reduced in an alkaline-cell battery?
9.
Which metal is used to coat iron in galvanizing?
10.
All of the following are the type of chemical bond exist in NH₄Cl. Except?
11.
The bond order of a homo nuclear diatomic molecule can be decreased by ______.
12.
In a typical multiple bond, the σ bond results from overlap of ______ orbitals and the π bond(s) result from overlap of ______ orbitals.
13.
Molecular Orbital theory correctly predicts Para-magnetism of oxygen gas, O₂. This is because ______.
14.
An anti bonding π orbital contains a maximum of ______ electrons.
15.
The molecular geometry and hybridization of ICl₄⁻ are
16.
Which hybrid orbital doesn’t contain unhybridized orbitals?
17.
The π bond in ethylene, H₂C=CH₂, results from the overlap of ______.
18.
Which of the following contains the correct types of bond that exist in phenol?
19.
Which of the following is incorrect about half-filled degenerate orbitals of phosphorus (P)? They have
20.
What are the four quantum numbers of 5f⁸ orbitals?
21.
If we compare the four quantum numbers of 3S¹ and 3S² orbitals the first three quantum numbers are the same except spin quantum number. This concept is more explained by ______.
22.
If the rate of decomposition of HI into its diatomic elements is 6.0 × 10⁻⁵ mol/L·s, what is the rate of formation of iodine in the same reaction?
23.
The rate law for a reaction is found to be rate = K[A]²[B], which of the following mechanisms gives this rate law
- I. A + B → E (fast)
E + B → D (slow)
II. A + B → E (fast)
E + A → C + D (slow)
III. A + A → E (slow)
E + B → C + D (fast)
24.
The de Broglie wavelength (in nanometer) of a particle which has a momentum of 13.25 × 10⁻²⁶ kg·m/s is:
25.
According to Faraday’s second law, the weights of different substances evolved by the passage of the same quantity of electricity are proportional to their ______.
26.
The preferential discharge of ions during electrolysis is influenced by the ______.
27.
A simple first order decomposition reaction is 50% completed in 0.693 s. How much time would it take for 90.0% completion?
28.
For the reaction A → B, the rate law is given by r = k[A]². Increasing the concentration of A by a factor of 3 will cause the rate constant to ______.
29.
What is the ratio of CH₃COOH/CH₃COO⁻, if the pH of the solution is 5 and the dissociation constant (Ka) of the CH₃COOH is 1.8 × 10⁻⁵?
30.
Addition of NH₄Cl to an aqueous solution of NH₃ cause which of the following to occur?
I. pH of the solution increase
II. pH of the solution decrease
III. Percent ionization of NH₃ decrease
IV. OH⁻ concentration decrease
V. pOH of the solution decrease
31.
The rate law of a reaction is rate = k[D][X]. The units of the rate constant are ______.
32.
Which ways of expressing concentrations is best for more dilute solution?
33.
The rate constants for a reaction at a variety of temperatures are shown in the table below. Calculate the activation energy of the reaction by using the data below. (R = 8.314 J/K·mol)
34.
An electron has a spin quantum number, mₛ = +1/2 and a magnetic quantum number, mₗ = +1. In which of the following orbital will it NOT be present?
35.
Of the following colligative properties, which is the most practical for determining the extent of protein aggregation?
36.
At 445°C, Kc for the following reaction is 0.020.
2HI(g) ⇌ H₂(g) + I₂(g).
A mixture of H₂, I₂, and HI in a vessel at 445°C has the following concentrations: [HI] = 2.0 M, [H₂] = 0.50 M and [I₂] = 0.10 M which one of the following statements concerning the reaction quotient, Qc, is true for the above system?
37.
Which of the following formula represent when soap reacts with hard water?
38.
Which of the following is true about the advantage of detergents over soaps?
39.
Which resonance structure of Cyanate ion (OCN⁻) makes the greatest contribution to the resonance hybrid?
40.
All of the following statements are correct about benzoic acid, **except**?
41.
What are the number of phases, components and degree of freedom for the mixture of white powders of sugar and potassium chloride are ______ respectively?
42.
Why higher carboxylic acids are almost insoluble in water? This is because
43.
The unit of Kp for the reaction
NH₄Cl(s) ⇌ NH₃(g) + HCl(g), is
44.
pKa values of three acids X, Y and Z are 5.7, 2.4 and 8.5, respectively. Which of the following represents the correct order of acid strength?
45.
Which of the following pairs **correctly** matches the name of polymers and their monomers?
46.
Which of the following have the same cell reaction as the electrolysis of dilute NaCl?
47.
Of the following ions, which has the smallest radius?
48.
Which of the following electrolysis used to produce NaOH solution?
49.
Which observation(s) reflect(s) colligative properties?
I. A 0.5 m NaBr solution has a higher vapour pressure than 0.5 m BaBr₂ solution
II. A 0.5 m NaOH solution freezes at a lower temperature than pure water
III. Pure water freezes at a higher temperature than pure methanol
50.
If we have three aqueous solution of Na₂SO₄ labeled as ‘A’, ‘B’, ‘C’ with concentrations 0.123 m, 0.0123 m and, 0.00123 m, respectively. The van’t Hoff factor for these solutions will be:
51.
Which one of the following acid-base definition classify BF₃ as acid?
52.
The ultra violet series that arise when higher energy electrons drop to the ground state is:
53.
Which one of the following is correct about the reaction between Lewis acid and Lewis base?
54.
Acetylsalicylic acid (Aspirin) has the structural formula:
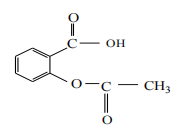
Which functional group (groups) is (are) present in this compound?
55.
If the equilibrium constant for the reaction
A + 2B ⇌ 4/3C + D
has a value of 10, what is the value of the equilibrium constant for the reaction 4C + 3D ⇌ 3A + 6B at the same temperature?
56.
An equilibrium that strongly favors products has ______.
57.
For which of the following elements is **Hund’s rule** used in writing the electron configuration?
58.
Which of the following equation is free from spectator ions?
59.
The main ore of Lead is:
60.
The pH of a solution prepared by mixing 50.0 mL of 0.125 M KOH and 50.0 mL of 0.125 M HCl is ______.
61.
What is the coefficient of the permanganate ion when the following equation is balanced? MnO₄⁻ + Br⁻ → Mn²⁺ + Br₂ (acidic solution)
62.
When 4.6 is divided by 12.786 the actual answer is 1.6511. What will be the correct answer?
63.
The IUPAC name of the following compound is ______.
64.
For a first order reaction, the concentration versus time of the reaction mixture is given by the following graph
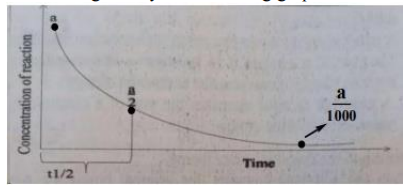
If a is the initial concentration of the reactant and its half-life is 0.1 hr., after how many hours would be the concentration of reactant produced to a/1000?
65.
Which of the following is Measuring equipment?
66.
The sanitizer is labeled 83.5% alcohol. How can one scientifically check the concentration?
67.
Who used “the cathode ray tube” to discover the electron and determine its charge to mass ratio?
68.
The unit of equivalent mass is ______.
69.
A. Conjugate base has one less H and one more minus charge than the acid.
70.
In which one of the following aqueous solution does molarity and normality are equal:
71.
Combining aqueous solutions of BaI₂ and Na₂SO₄ produce a precipitate of BaSO₄. Which ion(s) is/are spectator ions in the reaction?
72.
A 10.0 ml aliquot of HCl (aq) of unknown concentration was titrated with 0.1 M NaOH (aq). It took 100.0 mL of the base to reach the endpoint of the titration. The concentration (M) of the acid was ______.
73.
Sodium does not occur in nature as Na (s) because ______.
74.
Given the following reactions:
2S(s) + 3O₂(g) → 2SO₃(g) ΔH = -790 kJ
S(s) + O₂(g) → SO₂(g) ΔH = -297 kJ
Calculate ΔH° (in kJ) for reaction below, the enthalpy of the reaction in which sulfur dioxide is oxidized to sulfur trioxide
2SO₂(g) + O₂(g) → 2SO₃(g) is ______ kJ.
75.
Of the following, which one is a path function?
76.
A rock deposit that contains enough mineral to make it economically feasible to extract and purify to derive a desired product material is known as ______.
77.
During the extraction of aluminum by the electrolysis of molten aluminum oxide, what is the purpose of adding cryolite (Na₃AlF₆) to aluminum oxide?
78.
Which one of the following polymers is formed by condensation polymerization?
79.
Natural rubber is too soft and chemically reactive for practical applications. Vulcanization of natural rubber entails ______.
80.
The Kb of ammonia is 1.85 × 10⁻⁵. The pH of a buffer prepared by combining 75.0 mL of 1.00 M ammonia and 75.0 mL of 1.00 M ammonium nitrate is ______.