Welcome to your Chemistry national exam 2014
choose the best answer from given
alternatives
1.
What is the purpose of synthetic Cryolite in the extraction of aluminum by the Hall' s process?
2.
Which of the following is a chemical property of aluminum?
3.
How is tanning carried out? In tanning the hides or skins are
4.
How can a project of establishing an industry that consumes coal affect the environment?
5.
Which of the following is a fundamental quantity SI unit?
6.
What is enthalpy change? It is
7.
What is oxidation? Oxidation is
8.
What is meant by preferential discharge, if a number in a solution?
It means that last at given electrode.
9.
Faraday' s Second Law states:
10.
Which of the following is true about Zn- Cu voltaic cell?
11.
Which of the following postulates of Dalton' s atomic theory was modified by the modern atomic theory?
12.
Which of the following includes all of the known atomic models?
13.
The electron configuration of Cu( Z =29) is:
14.
what does Mendeleev's periodic law state ? properties of elements are periodic functions of their
15.
among the following reactions which one shows a coordinate covalent bond formation?
16.
Which of the following statements is correct regrading alkenes and alkynes?
17.
What is the general formula of alkynes?
18.
Which functional group containing compound is named using the suffix "- ol”?
19.
Which of the following represents the general formula of monotirdric alcohol?
20.
What is a Lewis acid? It is
21.
Which of the following statements is NOT correct about minerals and ores?
23.
Which of the following is NOT a major step in metallurgical processes?
24.
Which monomers are used for the synthesis of Teflon?
25.
Which of the following represents the structure of a disaccharide?
26.
What is the significance of electron probability distribution? It tells us about
27.
Which group in the periodic table contains elements with the valence electron configuration of ns2 n pp1 ?
28.
Which of the following is a correct Lewis structure for diazomethane ( CH2 N2 ) ?
29.
Which of the following is a property of covalent compounds? Covalent compound
30.
Which of the following is a covalent molecule?
31.
Which of the following carboxylic acids is a solid substance at room temperature?
32.
What is the IUPAC name of an ester having
the molecular formula C6 H12 O2 ?
33.
What is the formula of pentatonic acid?
34.
What is the rate of dissolution? It is
35.
Which one of the following is correctly stated regarding solubility as an equilibrium process?
36.
Which of the following assumptions of the kinetic molecular theory of gases explains the high compressibility of gases?
37.
Consider a Bottle containing water, water vapor and air in equilibrium. If the temperature decreases slightly, the amount of water vapors;
38.
Why do water droplets collect on the outside of a glass of water on a hot day?
39.
What are hydrocarbons? Compounds that contain
40.
Which of the following statement is INCORRECT regarding the boiling points of alkanes?
for the conversion of 3.S yards into meter using Answer: A od, which of the following is the correct step
41.
for the conversion of 3.5 yards into meter using Answer: A od, which of the following is the correct step
42.
In the scientific method, a predictive statement that can be tested experimentally ÎS:
43.
Which of the following is NOT true about cathode rays? Cathode rays
44.
Which of the following statements is correct about the energy of a photon?
45.
Which of the following explains the emission spectra of atoms consisting of a series of fine lines?
46.
What is the mole ratio of oxygen gas to carbon dioxide gas in the following reaction?
47.
Consider the following chemical equation for the combustion of ethane (C2 H6)in the presence of oxygen to produce carbon dioxide and water.
48.
Which of the following factors DOES NOT affect the rate of a reaction?
49.
How does a catalyst increase the rate of a reaction?
50.
Which of the following statements correctly describes Boyle‘ s Law?
51.
What is the concentration of a solution? It is
52.
How many moles of C12 H22O11(sucrose Molecular weight =342.91 / mol ) is needed there to prepare 2.0 L of 0.3M solution?
53.
At 75° C, the vapor pressure of pure water is 39kPa Which one of the vapor pressure of a solution obtained by dissolving 34.2 g sucrose in 180 mL of water? (Assume that the density of water is 1.000 g/mL )
54.
Which of the following statements is NOT true about the colligative properties of electrolytes and non-electrolytes?
55.
For the reaction shown below, which of the following is a conjugate acid - base pair ?
56.
What are intermolecular forces? They are forces due to the attraction between
57.
Which of the following belongs to chemical bonding theories?
58.
In which of the following molecules is the central atom sp2 hybridized?
59.
For a reaction:
1/2A -2&, the rate of disappearance of
' A' is related to the appearance of
' B' by the expression:
60.
The half life of a reaction is the time taken for the
61.
The self-ionization of water can be expressed by the equation:
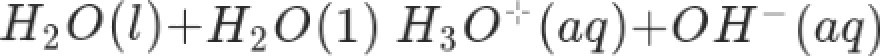
Which of the following is the correct
expression of the ionic product of
water?
62.
What is effect of a common ion in an electrolytic solution?
63.
Sodium carbonate is a basic salt because it is a salt of
64.
Which of the following statements is true for an acid-base titration?
65.
What is the change in internal energy ( AE) for a reaction, m which the system absorbs 650 J of heat and 125 J of work is done on the system?
66.
Which of the following is a polyprotic acid?
67.
Which of the following does represent a general property of a base?
68.
What are salts? salts are
69.
The process in which a compound absorbs electrical energy and decomposes into its element is
70.
Which of the following is true about galvanic cells used in commercial lead storage batteries ? in lead storage batteries.
71.
Which statement describes the formation of a covalent Bond? In covalent Bond electrons are
72.
How do the elements of Group IIA form ions?
73.
How is hydrogen bonding formed? is Fred to is bonded to
74.
Which one of the following forms of energy change occurs from the combustion of carbon in oxygen is
75.
What is the molar volume of a gas?
76.
In the first order decomposition of nitrogen pentoxide at 335 K
N2 O5 (g) --- 2N O2 (g)+1/2O2(g)
A reaction was carried out with 2.5 g of N2 O5 samples at
335 K and after 109 seconds, 1.50 g of N2 O5
remained
What is the value of the rate constant?
77.
What is the difference between reversible and irreversible reactions?
78.
The general formula for saturated monocarboxylic acids is
79.
The heating curve shown below shows a substance in the solid, liquid and gas phase. Which segment represents only the liquid phase?
80.
Consider the reactions all given bellow are in gaseous state. For which of the following
reactions IS KP less than KC