Welcome to your Chemistry national exam 2013
choose the best answer from the given alternatives
1.
The branch of chemistry that is involved in separation, identifying and determining the relative amounts of components in a sample of material is known as
2.
Significant figures are figures with
3.
For the determination of the density of a new ceramic, a student measured the mass of a piece on an analytical and obtained 3.8056 gram and its volume 2.5 mL by displacement of water in a graduated cylinder. The correct reported density of the ceramic should be
4.
The scientific method is
5.
One of the following is NOT a cause of uncertainty in measurement?
6.
Which of the fo8owlng is true about isotopes? Isotopes of an element have
7.
The following diagrammatic representations shows the shape for various orbitals. Which one represents the d2/z orbitals?
8.
Sliver (fi = 47) has several known isotopes, but two occur naturally, 107 Ag and 109 Ag. Given the following mass spectrometric data, calculate the atomic mass of Ag
9.
Which statement correctly describes Heisenberg's uncertainty principle?
10.
Which of the following statement is correct about energy changes when an electron changes its energy level?
11.
The sanitizer in wide use for protection against Covid-19 is labeled 83% alcohol. How can one scientifically check the concentration? This can be done by
12.
The correct electronic configuration for chromium (Cr, Z= 24) is
13.
According to Hund's rule equal energy (degenerate) orbitals are filled with
14.
In the modern periodic table
15.
The electrolysis of liquid (molten) NaCl in the set-up for the electrochemical cell is given in the following figure below
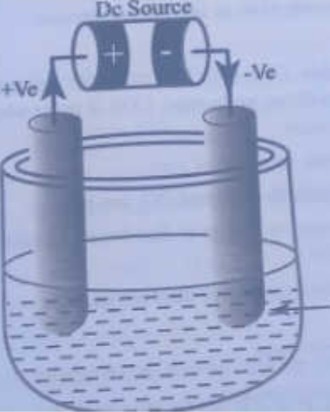
which of the following statement is true about the electrolysis of molten sodium chloride?
16.
Covalent compounds are liquids or gases at room temperature and have low melting and boiling points. Which of the fo8owing statements explains these properties of covalent compounds?
17.
Both carbonyl sulphide , COS(SCO), S= C=O and carbon dioxide (CO2)O=C= O are linear molecules. COS is polar whereas CO 2 is non-polar. this is because
18.
Which of the following statements is true about the formation of CH4 according to valence bond theory?
19.
Phosphorus ( Z = 15 ) has the electronic configuration 1s22s2 2p63s23p3 , what kind of hybridization does phosphorus undergo in the formation of phosphorus pentachloride(PCI5)
20.
One of the following reactions illustrates the formation of coordinate covalent bond?
21.
Hydrogen bonding occurs in compounds that contain H—N, H— O and H-F bonds. These bonds are stronger than the ordinary dipole-dipole interaction because
22.
Which of the following is an example of exception to the octet rule?
23.
Which of the following pair is true about the general properties of the different types of crystalline solids
Type of solid & Hardness & Melting point &
Conduction of heat of electricity
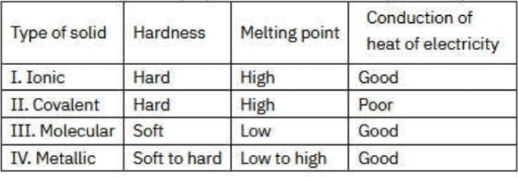
25.
Given the following catalyzed reactions
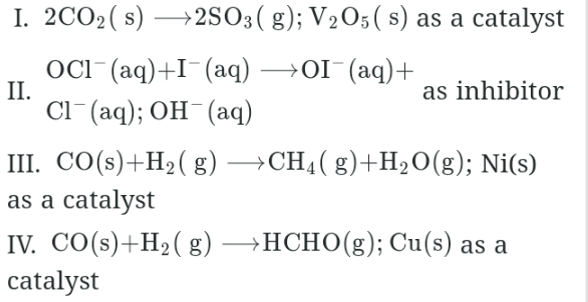
Which reactions is examples for homogeneous catalysis?
26.
Which statement is true about catalyst?
27.
According to the transition state theory
28.
Which of the following is true about the rate constant?
29.
For a first order reaction, the concentration versus time of the reaction mixture is given by the following graph
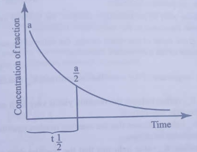
If a is the initial concentration of the reactant and if the half-life is l hr, after how many hours would be the concentration of reactant reduced to a/32
30.
The mathematical expression

Arrhenius equation is of important as it
shows
31.
From the magnitude of the contribution constant, Keq One can deduce that
32.
The statement "If a system at equilibrium is subjected to a stress, the system will readjust itself to reduce the effects of the stress is known as
33.
The law of mass action states that
34.
for the reaction between gaseous NO and O2 to form NO2 (g), what will be the equilibrium constant
35.
The equilibrium constant Kp for the reaction 2SO3 ( g)----- 2SO2 +O2 (g) is 8 X 10-3 Kp at 227° C.
What is the value of Kc for the reaction at
the same temperature?
36.
In the production of NH3 by the Haber
process according to the equation
N2 ( g)+3H2 ( g) 2NH3 (g) H=-92KJmo1- 3
What are the favorable conditions for the
production of high yield of NH3 ?
37.
Which one of the following statements describes laboratory and industrial preparation of acetic acid respectively?
38.
Which of the following is true about carboxylic acids?
39.
What is the IUPAC name for the branched carboxylic acid
CH3 — CH(C1) —CH(CH3) —CH(CH3) — COOH
40.
Esters are characterized by the following properties EXCEPT
41.
Oils are unsaturated fatty acids. The process of converting oils to solid fats involves?
42.
A chemistry teacher has added different reactants and reagents in three different test tubes as described below
I. Into the test tube A he added ethanol,
sodium dichromate, sulphuric acid
II. Into the test tube B he added
acetaldehyde, sodium borohydride and water.
III. Into the tube C he added methyl acetate
and ethanol and sodium hydroxide.
In which test tube would a reaction takes place that provide acetic acid?
43.
The difference between colloids and suspension is that
44.
The solubility of a substance is the amount of a substance that
46.
The "like dissolves like” rule accounts for one of the following observation
47.
Which of the following concentration units is correctly defined?
48.
The concentrated sulphuric acid H2 SO4 we use in the laboratory is 98% H2SO4 t by mass and has a density of 83 g/mL . Below are given the concentration of the acid in Varlous
units. Which of the given alternatives
correctly describes the concentration of the
acid?
49.
How do you prepare 500 mL of 1 NCa(OH )2 solution?
50.
The Ka of acetic CH3 COOH is 8 x 10-5 “
What is the percent ionization of
1MCH3 COOH ?
51.
According to the Lewis definition of an acid, an acid is a substance that
52.
Water is a weak electrolyte, because
53.
Which of the following reactions represent the amphiprotic behavior of water
54.
For an acidic solution, which of the following is correct?
55.
What is the pH of 0.254M HF solution at 25 C The ionization of the weak acid, HF is given by
56.
One of the following given pairs represents examples of buffer systems
57.
The reaction between a weak acid and strong base is presented by the reaction between acetic acid and NaOH gives a basic solution because
58.
An acid is a substance that
59.
One of the following given masses of acids constitutions the corresponding equivalent of the acid
60.
One of the following pairs shows Bronsted- Lowry acids behavior in water?
61.
In the reaction mixture of the two NH4 OH and NH4 Cl
62.
Which one of the following is correct about K w at different temperature? The ionic product of water Kw equals
63.
What is reduction? Reduction is
64.
During the electrolysis of dilute aqueous sulphuric acid, the ions present in solution
are H+ , OH+ and SO42- . Which ions are
preferentially discharged at the cathode and
anode, respectively?
65.
Which one of the following is NOT correct about electrodes and electrochemical cells?
66.
The mathematical expressions for Faraday's first law is summarized as
67.
In the electrolysis of copper sulphate solution using graphite electrodes, O2 is liberated at the anode and copper metal is deposited at the cathode. If the electrolysis of CuSO4 is performed using copper electrode, what happens at the anode and cathode?
68.
The statement "the amount of a substance consumed or produced in an electrolytic cell is directly proportional to the amount of electricity that passes through the cell' is known as
69.
Given the following electromotive series of selected metals with their standard reduction potentials
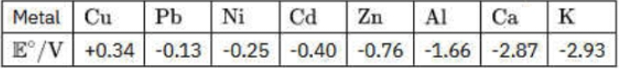
Which of the following statements is correct about reactivity of the metals?
70.
One of the following is NOT true about the effects of corrosion?
71.
in the Galvanic cell given in the diagram below
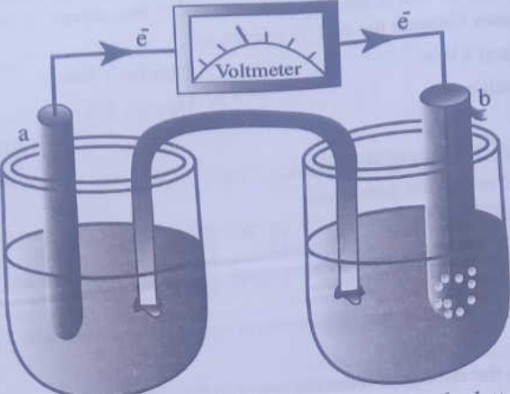
Identify the component parts correctly labelled for the letters a, b, c, d and choose the correct one among the following
72.
The conversion of nitrogen gas into useful nitrogen compounds is known as
73.
CO 2 is released into the atmosphere by one of the following processes
74.
One of the following is NOT a common application of silicon
75.
One of the following is NOT among the chemical properties of silicon
76.
Which one of the following is a common reaction of tin?
77.
The monomer of natural rubber is
78.
All of the following are examples of addition polymers EXCEPT
79.
One of the following examples of polymers is correctly matched with is uses.