Welcome to your Chemistry national exam 2012
Choose the best answer from the given
alternatives
1.
come major fields of chemistry that study all the elements and their known as
2.
Which of the following correctly defines accuracy of measurements?
3.
How many significant figures does a measurement of 0.00143 M 3 have?
4.
What is a scientific method? It is a method of
5.
A hypothesis step in the scientific method is
6.
Which of the following states the law of definite proportions?
7.
what did J.J. Thomson contribute with his experiments in cathode-ray tubes in understanding the atomic structure of the atom?
8.
An element x has two naturally occurring isotopes. x(10. 0l3amu) and X(11. 0l amu). The first accounts for 19.9 % of the total and the second accounts for remaining 80. 1%. What is the average atomic mass of the elements X ?
9.
What are isotopes? Isotopes are atoms with the same number of pro. tons but different number of
10.
Which statement is correct for the emission spectrum of the hydro gen atom?
11.
How much energy would be released as an electron is moved from the o=4 to the o 3 energy level?
12.
Which set of quantum numbers uniquely defines one of the electrons in an atomic orbital with n =2 and 1=0 ?
13.
What is Pauli Exclusion Principle? The Pauli Exclusion Principle states that
14.
Which of the following is the correct relationship between the given parameter and the property of an EMR?
15.
When equal amounts of sodium chloride and lead iodide are dis. solved in water, which of the following statements is true regarding the electrical conductivity of the dissolved ionic solution?
16.
Which of the following properties is true about covalent molecules?
17.
For the molecule (phosgene) shown below, why do the lone pairs and double bonds affect the bond angles?
18.
If you are provided with four covalent compounds namely naphthalene, graphite, iodine and ethanol to investigate their electrical conductivity, which one would you expect to conduct electricity either in the solid or in the liquid state?
19.
Based on the VSER model, what shape would you expect for the molecule [I3]-?
20.
Give the reaction:

The bond formed between NH3 and H+ is
21.
Which of the following is NOT the correct geometric configuration for the given molecule?
22.
Which one of the following substances has London dispersion forces as its only intermolecular force? (No hydrogen bonding, no dipole forces?
23.
Which of the follow ng is a basic chemical bonding theory?
24.
Which types of intermolecular forces are present in molecular crystalline solids?
25.
Which statement is NOT correct about the valence bond theory?
26.
Which of the following factors will affect the rate of chemical reaction?
27.
Which statement is correct for the reactions: 2 A+B --- C?
28.
The theory that states "the collision between two reacting species results in the formation of an activated complex" is known as
29.
For a reaction: A+B--C, the rate equation is r = k [A ] Which of the following is NOT correct?
30.
What is meant by rate-determining step in chemical kinetics?
31.
What is dynamic chemical equilibrium?
32.
The Reaction Quotient ( Q ) is
33.
What factors affect chemical equilibrium in a chemical reaction?
34.
Which of the following are equal for a chemical system at equilibrium?
35.
The equilibrium constant for the formation of calcium carbonate from the ions in solution is 2.2 x 103 according to the reaction:

What is the value of the equilibrium
constants for the reverse of this reaction?
36.
Which of the following explanation is correct about vapor pressure and its relation with phase changes?
37.
Which of the following structural formula represents propionic acid?
38.
Acetic acid is manufactured industrially by the fermentation of
39.
Which of the following DOES NOT represent an ester with the molecular formula of
40.
What is the characteristic smell of an ester?
41.
Which of the following statements is correct about fats and oils?
42.
Which of the following is the correct procedure during the preparation of soap?
43.
Which of the following descriptions about a suspension is correct?
44.
What is meant by the rate of dissolution of a solute?
45.
How can you prepare a supersaturated solution of sodium thievery :A if the solubility of the salt at 25‘ C is 50 g per 100 water?
46.
The solubility curves of some common salts in water at 30 C are shown below:
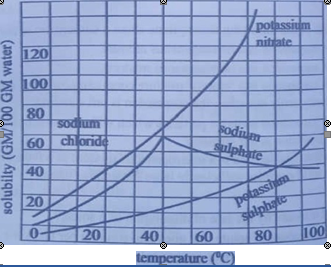
When 20 grams of sodium sulfate, Na2 SO4. is dissolved in 100 grams of water at 20‘C, the solution can be correctly described as
47.
What is mole fraction? Mole fraction is
50.
The thermal decomposition of potassium hydrogen carbonate yields potassium carbonate, carbon dioxide and water.
2KH CO3 MK 2 CO3 + H 2 O
How many moles of potassium carbonate can be produced from 550 grams of potassium hydrogen carbonate?
51.
Which of the following physical properties of a solvent are affected by the formation of a solution?
52.
A solution is made by dissolving 3.75 g of pure non-volatile solute in 95 g of acetone. The BP of pure acetone was observed to be 55.950 C and that of the solution was 56.50°c. Which of the following is correct based on the given evidence? If the dissolved solute is electrolyte. The boiling point of the solution
53.
In the following reaction, which is Bronsted -Lowry base?
54.
In the auto ionization of water
55.
Given the following acid-base reaction.
56.
What is the pH and [H+]of a 0.2M solution of HCN?
57.
A common ion effect in a certain weak acid will make the solution less acidic this is because the presence of a common ion
58.
A salt is formed from the weak acid (HCN) whose Ka—4.9 X 10- 10 and a weak base whose Kb 1.8 X 10 5. What do you expect about the solution of the salt formed from HCN and NH3
59.
Thee titration curve shown below shows the titration of
60.
What is the difference between equivalence point and end porn acid base titration?
61.
Which one of the following does a closed system allow to happen?
62.
The standard enthalpies of formation of HBr (g ) and Br2 s) are -36 and +31 k J moll —1 (at 298K), respectively. What is the enthalpy change, DH for the fo8owing reaction?
63.
Which of the following statements about the standard state of a substance is correct?
64.
Which of the following statements best describes the Second Law of Thermodynamics?
65.
Colligative property is defined as a property that depends on
66.
Which of the following represents a net ionic equation?
67.
Which of the following statements is correct about the solution process?
68.
The number of equivalent per liter of solution is known as
69.
When silver nitrate solution is mixed with sodium chloride solution it gives an insoluble silver chloride. Which of the following correct?
70.
Which of the following explains the change of colligative properties of a solvent upon addition of a non-volatile solute?
71.
If 3.423 grams of source is dissolved in 200. 0 g of water, what is of molarity of the sucrose solution? (molar mass of sucrose =342.3 V mol)
72.
What is the Arrhenius concept of a base?
73.
The Bronsted Lowry concept defines an acid as a substance that
74.
Which of the following explains the cleaning action of soap ?
75.
Which of the following statements is WRONG about entropy and change of entropy ( A )
76.
Given a general reaction at equilibrium: aA+bB nM +nN The equilibrium constant ( kc) expression is
77.
The first law of thermodynamic states that
78.
Given the population of ammonia.
79.
Which of the follow is a homogeneous reaction?
80.
Which of the following procedures indicates a system which CANNOT exchange both energy and matter with the surroundings?