Welcome to your Chemistry national exam 2011
1.
The oxidation state of chlorine in HClO4 is
2.
Which substance is used to lower the melting point of aluminum oxide ore in the electrolytic extraction of aluminum?
3.
The metal extracted from limestone, chalk and marble is
4.
Which one of the following is NOT a condensation polymer?
5.
Which of the orbitals in the figure below has (have) an angular momentum quantum number of 1=2 ?
6.
Which of the following compounds does NOT contain both ionic and covalent bond?
7.
What does it mean by AH- negative for a given process?
8.
Which of the following can function as both a Bronsted- Lowry acid and Bronsted - Lowry base?
9.
What makes the phosphorus cycle different from the carbon and nitrogen cycles?
10.
In the electromagnetic spectrum with wavelength shown (in micrometers,μm ), which bracketed section of the spectrum represents visible light?
11.
What is the first step in a scientific investigation?
12.
Which of the following is the SI measurement unit of electric current?
13.
What aspect of the modern view of atomic structure was proved by Rutherford s gold foil experiment?
14.
Acetylsalicylic acid (aspirin) has the structural formula:
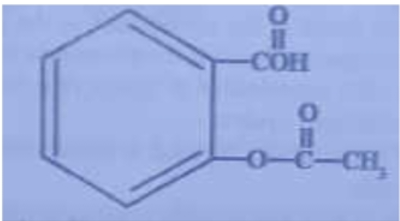
Which functional group (groups) is (are)
present in aspirin?
15.
which of the four colligative properties arises in systems where there is an equilibrium between a liquid solution phase and a second liquid phase?
16.
When Na2 CrO4 t solution is acidified, which of the following is formed?
18.
Which of the following is NOT a component of polysaccharides?
19.
Fats and oils can be classified as
20.
The molar equilibrium concentrations for the reaction mixture represented above at 298 K are [K ] = 40M, [M]=5 .0M, and [Z] 2.0M. what is the value of the equilibrium constant, K eq for the reaction at 298 K ?
21.
What is the IUPAC name for the following carboxylic acid?
22.
4 L of 0. 02M of aqueous solution of NaCl is diluted with 1 L of water. What is the molarity of the solution?
23.
How much water, in liters, must be added to 0.50 liter of 6.0MHCI to make the solution 2.0M ?
24.
The pH of a solution prepared by the addition of 100 mL0.002M HCl to 100—mL distilled water is closest to:
25.
What is the pH of a 0.10M solution of NH3 , a weak base, whose Kb =1.8 x 10-5?
26.
There is a progressive decrease in the bond angle in the series of molecules CC I4 Cl 3, and H2O according to the VSEPR model, this is best explained by:
27.
The appropriate unit for a first order rate constant are?
28.
Consider the following two possibilities for electron transfer in a hydrogen atom, given below:
First: The electron drops from the Bohr Orbit
n =3 to the orbit n= Followed by the
transition from n=2 to n=1
Second: The electron drops from the Bohr orbit n=3 directly to the orbit n=1
Which of the following is correct about the energy change these transitions?
29.
Which of the following elements has the highest fifth ionization energy (IE5) ?
30.
When a saturated solution of sodium chloride is heated, it becomes
31.
Hydrolysis of ester leads to the formation of which of the following products in basic medium?
32.
A sealed isothermal container initially contained 2 moles of CO gas and 3 moles of H2 gas. The following reversible reaction occurred:
CO(g)-F2H2 (g)---- OH3 OH(g)
At equilibrium, there was I' mole of CH3 OH in the container. What was the total number of moles of gas present in the container at equilibrium?
33.
Which groups in the periodic table form ionic bonds?
34.
Which of the following is the correct reaction taking place at the electrodes during the electrolysis of dilute sodium chloride solutions?
36.
Which one of the following type of bonding exists between atoms with very different electro negativities?
37.
Considering only resonance structures that are major contributors to the overall bonding in PF5 , which of the following statements is correct?
38.
Which one of the following reactions at equilibrium would be unaffected by an increase in pressure?
40.
Two types of polymer are shown below. Which of the following statements concerning these polymers is correct?
41.
The energy of an electron m the first Bohr orbit of hydrogen atom is —13.6eV. The possible value of the excited state for electron in Bohr orbits of hydrogen is
42.
The sublevel that can be occupied by a maximum of 10 electrons is identified by the letter
43.
.
What is the molecular shape of ICI4- ?
44.
A sample of solid ammonium carbonate is heated in a closed container at 298 K and allowed to reach equilibrium.
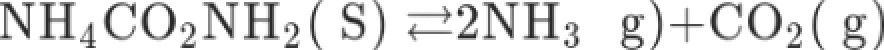 If the total pressure of the system is 0. 114 Citi»
If the total pressure of the system is 0. 114 Citi»
, what is the value of equilibrium constant,
Kp ?
45.
What hybridization change, if any, occurs at the underlined atom in the following reaction?
46.
Based on the information given in the table below, what is the enthalpy change for the reaction:
47.
Which of the following Reactions is NOT involved in the contact process, during the production of sulfuric acid?
48.
In which of the following cases will the dissolution of sugar be the most rapid?
49.
How many grams of iodine , /2› must be dissolved in 225.0 mL of carbon disulfide, CS2 (density =1 .261g/cm3) , to produce a 0.11G iii solution?
50.
The minimum energy required for an effective collision is called
51.
Which of the following statements is correct about nitrosyl chloride (NOCI) ?
52.
Which of the following is NOT true about the photoelectric effect?
53.
A chemist creates a buffer solution by mixing equal volumes of a 0.2 molar HOCI solution and a 0.2 - molar KOCI solution. Which of the following will occur when a small amount of KOH is added to the solution?
I . The concentration of un dissociated HOCl will increase.
II. The concentration of one ions will
increase.
III. The concentration of H^ ions will
increase.
54.
The proposed reaction mechanism between nitrogen monoxide and bromine is give below.
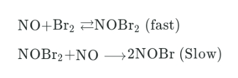
Which of the following rate equation is consistent with the proposed mechanism?
55.
Which one of the following statements regarding a dynamic equilibrium is FALSE?
56.
If a solution of acetic acid (CHPCOOH) has a pH of 3.00 , what is its concentration? Ka of acetic acid =1. 74 x 10-5
57.
The indicator methyl red is red in a solution of NaH2po4 Which of the following equation is consistent with this observation?
58.
A nickel electrode can undergo oxidation to Ni2+ ion as follows:
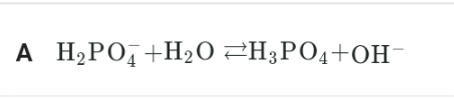
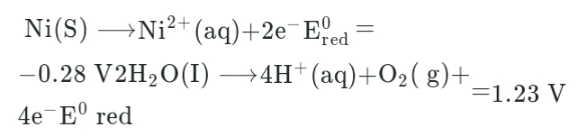
What will happen during the electrolysis of concentrated aqueous solution of NiSO4 using Ni electrodes?
I) Nickel will deposit at the cathode.
II) Nickel will dissolve at the anode.
III) Oxygen will be released at the anode.
IV) Nickel will deposit at the anode.
59.
features common to both galvanic and electrolytic cells include which of the following?
I. Oxidation at the anode
II. Can perform electrolysis
III. Spontaneous
60.
Which of the following has the same number of significant figures as the number 1.00310 ?
61.
When an electron in a hydrogen atom makes the transition from the n=4 state to the n-2 state, blue light with a wavelength of 434 nm is emitted. Which of the following expressions gives the energy released by the transition?
62.
Arrange the following molecules in the order of increasing stability.
63.
The reaction below takes place with all of the reactants and products in the gaseous phase. Which of the following is true of the relative rates of disappearance the reactants and appearance of the products?
64.
Which of the following metals cannot be electroplated on to the surface of another metal using an aqueous electrolyte?
65.
In the figure shown below, what does O denote?
66.
If 49 grams of H2 SO4 react with 80.0 grams of NaOH, how much reactant will be left over after the reaction is complete?
67.
which one the following is true?
68.
A certain current produces 0.50 g of hydrogen gas in 2.0hrs. What is the amount of copper liberated from a solution of copper sulfate by the same current flowing for the same time?
69.
For which of the following half-cells is the reduction potential E independent of the pH of the solution?
72.
Which of the following compounds is the least soluble in water?
73.
During esterification of carboxylic acid with alcohol which bond of carboxylic acid undergoes cleavage?
75.
Consider the following equation
76.
What is the basis for the scientific method?
77.
Which of the following molecules has the largest dipole moment?
78.
In three different experiments the following results were obtained for the reaction A products:
79.
What is the change in internal energy of a system that releases 12.4J of heat and does 4.2 of work on the surroundings