Welcome to your CHEMISTRY ENTRANCE 2016
1.
what are molecular formulae of an alkene and alkyne containing seven carbon atoms, respectively?
2.
Which of the following is the CORRECT method of preparation of alkenes in the laboratory?
3.
Which of the following is the CORRECT structure of benzene?
4.
Which of the following is the product of fractional distillation of crude oil?
5.
What is the molecular formula and IUPAC name of a saturated monocarboxylic acid having six carbon atoms?
6.
Given the following structure:
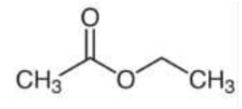
What is the IUPAC name for the above
structure?
7.
During summer, the average value for the temperature measured in a certain chemistry laboratory is 298.15 K . How many decimal places are there in the measured value?
8.
Which of the following is the CORRECT Lewis electron dot symbol of MgO ? (Atomic number: Mg =12 and O =8 )
9.
A student collected four beakers in his/her laboratory and added some small amounts of the following: naphthalene to the first, graphite to the second, iodine to the third and alcohol to the fourth. If they added equal volume of water to each of the above beakers and shake each beaker, which of the following will be their observation?
10.
Which of the following explanations about reversible and irreversible reactions is CORRECT?
11.
Consider the following three steps:
Step: 1 Electrolysis of water and fractional distillation of air
Step: 2 Passing hot mixture of gases
through a condenser
Step: 3 Introducing hydrogen and nitrogen gases in a chamber containing iron particles at a temperature of 300—500°C and a pressure of 15—25MPa
Which of the following is the CORRECT sequence involved during the industrial production of ammonia using the Haber process?
12.
Which of the following is a physical property of nitric acid, HNO3?
13.
Which of the following is the CORRECT explanation about herbicides?
14.
A student collected the following information from the community on the preparation of the local alcoholic drink "ARAKI":
Step: 1 Distillation of the liquid mixture in traditional ways
Step: 2 Adding a proportional amount of water to liquidity the tick dough - like mixture and leaving for 1-2 days for further fermentation
Step: 3 ”Bikel" preparation from barely, the starter ("Tinses”) preparation by mixing the Bikel with Gesho tRhamnuspronoides) powder and fermentation of the starter for few days
Step: 4 Baking of bread from different ingredients and breaking down the bread and mixing with the fermented starter and leaving for fermentation for couple of days
Which of the following is the correct procedure for the preparation of Araki
15.
Which one of the following synthetic polymers is used to make squeeze bottles, plastic wrapping and electrical insulation?
16.
Which of the following descriptions of the property of a covalent compound is CORRECT?
17.
The attractive force between molecules is known as
18.
The hybridization of the central atom xenon ( Xe ) in xenon tetrafluoride , XeF4, is p3d2. Which of the following is the shape of XeF4
19.
Which of the following is the CORRECT electron configuration of a peroxide ion, O22-
20.
Consider the following reaction between
nitrogen (N2) and oxygen (O2)
chollowing reache N2( g)+O2(g )→ 2NO (g)
the rate of disappearance of N2is 2.5x 10M/s, what is the rate of reaction for the formation of NO ?
21.
Which of the following is a Lewis acid?
22.
The pH of a 0.10 M solution of an aqueous solution of a certain acid is 3 . What is the value of acid ionization constant (Ka) of this acid?
23.
The shift in the position of equilibrium caused by the addition of an ion already involved in the reaction is known as
24.
Which of the following is CORRECT about equivalents of acids and bases?
25.
A 250 mL solution is formed from 24.5 g of sulfuric acid (IH2SO4). What is the normality of this solution? (ML. wt. H2SO4 =98 g/mol )
26.
Which of the following reaction is used for the preparation of bases?
27.
Which of the following salts is used in the treatment of waste water?
28.
A student prepared two beakers, each containing 100 mL of water. The student dissolved 10 mL of concentrated HCl in the first beaker and 10 mL . of concentrated CH3COOH in the second beaker, which of the following would occur in the solution?
29.
Consider the following five steps during the extraction of aluminum from is baunite ore by the Hall process:
I. Treating the mixture of an aqueous solution of sodium silicate and aluminate with an acid
II. Heating aluminum oxide strongly in a
furnace
III. Heating the ore with sodium hydroxide
solution
IV. Conversion of aluminum oxide to soluble sodium aluminate
V. Electrolysis of molten mixture of cryolite and aluminum oxide Which of the following is the CORRECT sequence of production
30.
Which of the following food preservation methods leaves a product without loss of aroma or flavor?
31.
The electronic configuration of an element ln the periodic table is
1s22s22p63s23p64s23d104p5.In which block does this element belong?
32.
Consider the following chart of general periodic properties in the periodic table:
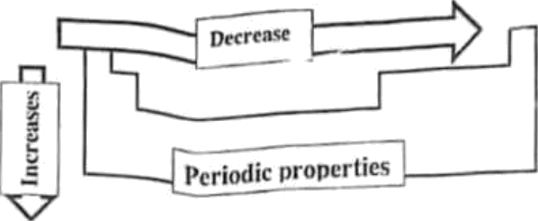
From the given periodic properties, which one CORRECTLY agrees with the periodic trend in the chart?
33.
Which of the following is CORRECT about the formation of a covalent bond? A covalent bond is formed
34.
The molecule of carbon tetrachloride ( CCI4) has four polar ( C-CI) bonds. However, CCI4 is a non-polar molecule. Which of the
following explains the reason for the
observed property of CCI4?
35.
Which of the following is responsible for the unusual high boiling points of HF, H2O and NH3
36.
from the assumption of kinetic molecular theory of gases, which of the following is CORRECT?
37.
Which of the following is CORRECT about the phase change observed ” water?
38.
Given the symbols of the three subatomic particles; electrons (e ), proto (p+) and neutrons (nº) which of the following is the
CORRECT compare's: of the absolute masses of protons, electrons and neutrons?
39.
In the modern periodic table, what is the name of the group that contains the most electronegative elements?
40.
A given element has atomic number of 55 and mass number of 133. Which of the following describes CORRECTLY the property of this element?
41.
Consider the mechanism for the of
N2O5 TO NO2:
First step: 2N2O5(g)

42.
Consider the following reaction:
N2(g)+O2(g)⇌NO(g)
The equilibrium concentrations of gaseous nitrogen, oxygen and nitrogen monoxide in a sealed container are as follows: [N2]= 1x 10-M,[O2] =2x 10-5M and [NO]=4X10-5M. What is the value of the equilibrium constant for the reaction?
43.
At a certain temperature the following reaction has an equilibrium constant KC= 12.4.
N2(g)+3H2( g)-2NH3(g)
if [N
2]=2.0M,[H
2]=2.0M and [NH
3]=10.0M,to which direction will the reaction proceed to reach equilibrium ?
44.
Which of the following is the general formula for saturated monocarboxylic acids? ( R is an alkyl group)
45.
Upon standing long, "Tella" or "Tej" turns sour. Which of the following is the CORRECT explanation for the observed effect?
46.
A student is interested in determining the content of a leaf from his/her environment and performed the following.
Step; 1 He/ she dried the leaves up in sun
light
Step: 2 Evaluated the work and draw conclusions
Step: 3 Collected leaves from the tree around his village
Step: 4 He/ she boiled the leaves and collected the leaves extracted
Step: S Analyze the content of the leaves extracted using an instrument
Which of the following is the CORRECT procedure if a student developed an experimental skill?
47.
Which of the following characteristics of an electromagnetic radiation is CORRECT?
48.
The hydrogen atom moves in a fixed circular orbit associated with allowable energy states”. This is called
49.
Which of the following is the core electron configuration of the metal ion in ferric sulfate, P2(SO4)? (Atomic Number of Fe =26)
50.
A chemical bond that results from the attractive force between shared electrons and nonmetal nucleus is called
51.
Which of the following is peroxide?
52.
Among the following which one is a ternary acid?
53.
A student collected few lerrions from his/her backyard, brought it the their class and prepared a lemeon juice in a beaker. They got blue and red Fiume papers from his/her teacher and placed few drops of the lemon juice both on the red and blue litmus papers. What will be their observation on the litores papers?
54.
What is the pOH of pure water at 37°C, where K2 equals 2.50 x 10-14 at its pH is
6.8?(log25 =0.4)
55.
The hydrogen ion concentration in a certain solution at 25º C is 1.0 x 104 M . What is the pOH of this solution?
56.
Given the following oxidation - reduction reaction:
H2S+HNO3→S+NO+H2O
Which of the following is CORRECT from the
given equation?
57.
Consider the following diagram of an electrolytic cell:
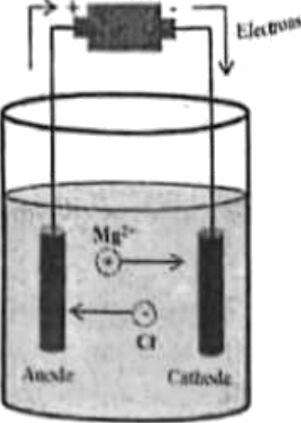
Which of the following is CORRECT from the given diagram?
58.
Which of the following is INCORRECT about molten electrolytes and aqueous electrolytes?
59.
Which of the following is NOT the industrial application of electrochemistry?
60.
Which of the following chemical industries is INCORRECTLY matched with its products?
61.
A student added 25 mL of AgNO3 solution to a beaker and weighed the mass of the beaker and its content as m1. He/she added 50 mL of a saturated solution of NaCI into the second beaker and weighed the mass of the beaker and its content as m2. Finally, he/she mixed the two solutions and weighed the mass of the resulting solution with the two beakers and recorded as m3.If he/she found that m3=m1+m2 which one of the following laws is investigated by this trial?
62.
A chemical reaction that releases heat energy to the surrounding is known as
63.
A type of reaction in which two compounds react together to form two or more products by exchanging reactants ions is known as
64.
"At a given temperature and pressure, equal volumes of different gases contain equal number of molecules”, this is known as
65.
Which of the following includes the preconditions required for a chemical reaction to occur?
66.
Consider the following reaction of zinc carbonate, ZnCO3:
ZnCO3(s)→ZnO(s)+CO2(g)
The chemical reaction that takes place during the conversion of ZnCO3 is classified as
67.
CaCO3+HCI→CaCI2+CO2+H2O(unbalanced)
if 40 g of CaCO3 reacts with 50 g of HCI, which substance is limiteing and which is the excess reagent ? (Atomic mass
Ca=40 g/mol, C=12g/mol
O=16g/mol,H=1g/mol and CI=35.5g/mol)
68.
A sample of a certain gas has a volume of 100 L at 27°C and 750 tor. The gas is expanded to a volume of 250 L at 200 tor. What is the final temperature of the gas?
69.
Which of the following is a CORRECT explanation of the given term?
70.
Which of the following is the general formula of alkenes?
71.
What are the two types of polymerizations?
72.
Which of the following is a monomer of natural rubber?
73.
The component of the environment consisting earth's upper outer most skin, and accessible to human beings is known as
74.
Consider the following steps that occur during the nitrogen cycle:
Step: 1 Reduction of nitrate by microbial
action
Step: 2 fixation of nitrogen by bacteria and
algae
Step: 3 Nitrification by nitro somas and nitrobacteria
Step: 4 DE nitrification of nitrate with acid and form ladyde
Which of the following is the CORRECT sequence in the nitrogen cycle?
75.
Some hazardous substances damage and create unwanted changes in air. water, soil or any other natural resource. The resulting change created is known as:
76.
Given the following heterogeneous reaction:
CaCO3(s)≃CaO(s)+CO2(g)
What is the equilibrium constant expression for the above reaction?
77.
Which of the following statements is CORRECT regarding the properties of monocarboxylic acids?
78.
What is the scientific notation for 0.0000055 ?
79.
A water molecule has two bond pairs and two lone pairs of electrons. Which of the following is CORRECT about these pairs of electrons?
80.
Which of the following CORRECTLY describes conjugate acids and conjugate bases?