Welcome to your CHEMISTRY ENTRANCE 2015
1.
In a kinetic experiment, a student placed crystals of iodine in a closed reaction vessel, introduced a given quantity of hydrogen gas, and collected data to calculate the rate of formation of hydrogen iodide. What would be the effect obtained on the rate of the reaction, if the student takes more amount of iodine from that used in the first experiment?
2.
Which of the following CORRECTLY relates the vapor pressure of a liquid with its boiling point? Boiling point is
3.
Consider this equilibrium system:
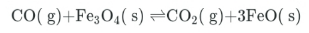
Which of the following disturbances shift the equilibrium position to the right?
4.
Which one of the following resulted from interaction of the atomic orbitals of bonding atoms to produce new electron distribution associated with the entire molecule?
5.
Which type of organic substances would be produced by heating a mixture of a carboxylic acid and an alcohol in the presence of H2SO4?
6.
Which one of the following is the reducing agent in the following reaction?
7.
Carbon forms two stable compounds with and carbon dioxide carbon monoxide carbon monexion of carbon, the ratio of oxygen in carbon monoxide to oxygen in carbon dioxide is 1 :2. This result is consistent with the law of
8.
When a system at equilibrium is disturbed by a change of variable, the system shifts the equilibrium position in a way that tends to counteract this change of variable. This is a statement for
9.
Which one of the following signs illustrates the rate change in terms of change in reactant concentration with time?
10.
Which one of the following gas laws states the volume of a fixed amount of gas mammancel al constant pressure is directly proportional to the absolute temperature of the gas?
11.
The PH of 0.1 M solution of formic acid (HCOOH) is 3 . What is the Ka of the acid?
12.
A system does 15.1 of work and no other changes take place. Which of the following function has fallen by 15 ?
13.
Which of the following is CORRECTLY stated about the spontaneity of a reaction?
14.
In thermodynamics: solutions in a beaker, a gas in a cylinder, a reaction vessel, a biological cell are examples of a
15.
Given the following: bond energies ( kJ/mol )
17.
Which type of substances has the following general formula? ( R1,R2 and R3 hydrocarbon groups.)
18.
The rate law for this reaction
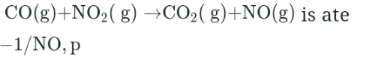
Which one of the following could be a plausible mechanism for reaction?
19.
The speed with which a solute goes into solution is
20.
Which one of the following explains how 2 L of I M Na OH aqueous stock solution is converted into a 0.25 M Na OH solution?
21.
Which one of the following IS TRUE about isotopes?
22.
Which one of the following describes the quantum mechanical model? The locations of electrons in an atom are described in terms of probability.
23.
Which one of the following is the diagrammatic representation of 35CI
17
24.
Which one of the following is an ionic compound?
25.
An element is located in Group VIIA and 3 rd period of the modem periodic table. On the basis of this the element;
26.
Which of the properties of solvents WOULD NOT he affected by the formation of a solution with a non-volatile solute?
27.
The partial pressure of acetylene, C2H2,in a container is 10 atm at 25°C. What is the solubility in molarity of C 2H2, assume that C2H2 obeys Henry's
:law?(kH =1.04 mol/Latm)
28.
What is the normality of a solution that contains 19.6 g of H2SO4in 20 mL of the solution?
29.
Which one of the following illustrates an extensive property?
30.
The point in a titration at which the number of moles of OH ions added to a solution is equal to the number of moles of H ions originally present is
31.
Which one of the following is TRUE about the electrolysis of a mortem hypothetical electrolyte, MX?
32.
During the extraction of an unknown metal, the metallic ore was fused with NaOH in air and then dissolved in water to obtain the soluble unknown metal containing species. Reduction with carbon is followed after the solution is treated with HCl . Finally the obtained oxide is reduced to its metallic form by aluminum. The most probable unknown metal would be
33.
Why tin does NOT react with cold water but calcium reacts with cold were quite readily?
34.
Why is vulcanization performed in natural rubber
35.
Which one of the following explains the determination of the cell potential of Zn—Cu cell using voltmeter?
36.
Which one of the following is an example of molecular crystals?
37.
Which one of the following COULD NOT be considered as factor influencing the rate of chemical reaction?
38.
Which one of the following shows the hybridization scheme of the central atom in H2O 2
39.
In which of the following order of a reaction, a plot of in [A]1 versus time is a straight line whose slope is - k ?
40.
A forward and reverse reaction continues to take place at equal rate in microscopic level without change in composition at macroscopic level. This statement is the definition of
41.
The heat change at constant pressure that results when 1 mole of a compound is formed from its elements in their standard states is
42.
What is a redox reaction? It is a reaction in which
43.
Which one of the following explains how electroplating takes place?
44.
How does the equivalent mass of a base is obtained?
45.
Which one of the following reactions would occur spontaneously in an aqueous solution at 25OC ? Assume that the initial concentrations of the dissolved species are all 1.0 M .
46.
Which of the following statements describes chemical industries? It is
47.
What will be the product if phosphorus reacts with excess amount of oxygen?
48.
One of the basic differences between metallic conduction and electrolytic conduction is:
49.
Which one of the following gases is released from combustion of a fuel in an industrial machinery causing global warming?
50.
The number of digits to the right of the decimal point denotes:
51.
Which of the following would occur during the formation of ions?
52.
In order to illustrate the structure and properties of a solid, a student put hundred marble balls in to a box that representing metal cautions and the empty space between and around the marble representing the electron sea. Which one of the following bonding is represented by this model?
53.
Which one of the following explains dipole-dipole force?
54.
Which one of the following reactions creates energy in our bodies during respiration?
55.
A reaction that involves the exchange of positive and negative ions of each reactant". This is a definition of a
56.
What are the four quantum numbers for an electron in a 2𝑠 orbital?
57.
A student uses a digital balance (±0.02 ), a thermometer(±0.1), a measuring cylinder (±0. 05 ), and a beaker (±0.5 ) during an experiment. Which of the above instrument is more precise?
58.
Which one of the following explains that light has BOTHA wave and particle nature?
59.
A nuclear breakdown in which particles or electromagnetic radiation emitted is
60.
Which statement describes valence shell electron pair repulsion (VSEPR) theory?
61.
Which one of the following is a colorless, flammable volatile liquid and miscible with non-polar solvents?
62.
The IUPAC name of the compound with the following structure is:
63.
Which one of the following petroleum products is used as a solvent and in dry cleaning?
64.
Which one of the following in NOT TRUE about laboratory preparation of ethylene by dehydration of ethanol?
65.
Which one of the following is an essential nutrient of plants?
66.
Which one of the following is the main reason for the large difference in dipole-moment between the triatomic molecules water (H2O) and carbon dioxide (CO2) ?
67.
How many non-bonding pairs of electrons are present in the valence shell of the central atom in CIF3 ?
68.
Which one would be observed in a laboratory activity that is applied on naphthalene?
69.
Which one of the following statements is CORRECT?
70.
The energy required to completely separate one mole of a solid ionic compound into gaseous ions is a/an
71.
Which of the following compounds reacts with basic oxide to form salt and water?
72.
Which one of the following reaction represents formation of an acid from its salt?
73.
The negative logarithm of the molar concentration of hydrogen ion to the base ten is known as
74.
Which one of the following galvanic cell is an example of secondary cell?
75.
Which one of the following is responsible for electrolytic conductivity?
76.
Which one of the following statement is TRUE about gases?
77.
Which one of the following mathematical expressions is derived from the combination of Boyle's law, Charles’ law and Avogadro's law?
78.
180 g of calcium carbonate (CaCO3)is allowed to react with 156 gram of hydrochloric acid (HCI), according to the following reaction. Which of the following substances is the limiting to thent?
(Atomic masses)
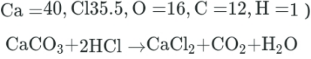
79.
Which one of the following isomers differs in the arrangement of carbon atoms in the longest continuous carbon chain?
80.
Which one of the following methods can be used in the laboratory for the preparation of alkynes?