Welcome to your CHEMISTERY GRADE 8 MODEL EXAM 2014
CHEMISTERY GRADE 8 MODEL EXAM 2014
Choose the correct answer
1.
Which of the following chemical product is produced at Zeway in Ethiopia?
2.
A pure substance that CANNOT be broken down into simpler substance by an ordinary chemical reaction is called
3.
A change that results in the formation of new substance with its own property is known as
4.
Which of the following apparatus is used for the separation of salt from salt solution?
5.
What is the name of an element having the chemical symbol “Ag”?
6.
The combining power of an element is called
7.
What is the formula name of CuO?
8.
The statement “In a chemical reaction the total mass of the reactants is equal to that of the products’’ is the law of
9.
Which of the following statement is true about Dalton’s atomic theory?
10.
What is an energy level (atomic shells)? It is the
11.
The lightest sub - atomic particle is
12.
Which one of the following is poly atomic molecule?
13.
The vertical column of the elements in the periodic table is known as
14.
What is the name of an alkene that contains five carbon atoms?
15.
One of the following is NOT used for identification of substances.
16.
Which one of the following is not chemical industry?
17.
Which of the following is a plant nutrient?
18.
Which of the following generally decreases from left to right across a given period?
19.
The group or elements with higher ionization energy as compared to other groups are:-
20.
Which one of the following is the effect of global warming? It
21.
Which one of the following is the application of chemistry in agriculture?
22.
Which of the following statement is true about the difference between elements and compounds?
23.
Which of the following methods of separation is used to separate a mixture of salt and water?
24.
The coefficient 3 in the formula 3CI2represents
25.
In balancing a chemical equation using the least common multiple (LCM) method the following steps are followed.
26.
Which of the following is the correct step for balancing a chemical equation using the LCM method?
27.
Which of the following statements describes the short comings of Dalton’s atomic theory?
28.
What is the maximum number of electrons that the L-shell can accommodate?
29.
If an element has an electronic configuration of 2,8,1, to which group does it belongs in the periodic table?
30.
Given the following chart that shows portion of the periodic table
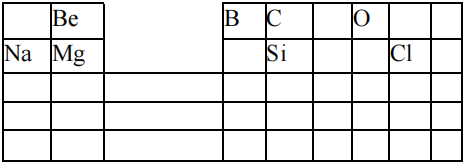
Which of the following statement is true about the trends in properties of the elements in the chart?
31.
One of the following is a property of acids. Which one is it?
32.
Which of the following element is found in free elemental form in nature?
33.
Which one of the following elements CANNOT be found free in nature?
34.
Oxygen mainly occurs in the form of
35.
Which of the following is NOT true about the occurrence of sulphur?
36.
Which water treatment method uses micro-organisms in water purification?
37.
Which one of the following natural fertilizer is used to improve soil fertility?
38.
Which of the following is the use of coal? It is used
39.
The first step in determining molecular mass of a substance is
40.
In the table below the physical properties of certain elements are given.
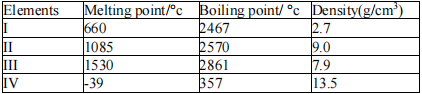
Which of the following is true about the elements? Element
41.
Which of the following is a molecule of an element?
42.
To which group and period of the periodic table does an element with atomic number 11 belong respectively?
43.
Which one of the following oxides is acidic?
44.
Given the following incomplete chemical equation. Na(s) + H2O(1) —? What are the products of this reaction?
45.
Among the following, which one is a binary salt?
46.
Which one is an alloy that made up of copper and zinc?
47.
If in a certain laboratory dilute nitric acid is added in to a beaker containing ammonium hydroxide, what would be the product?
48.
What is the molecular mass of white phosphorus, P4?
(Atomic mass: P = 31)
49.
Which property of element decrease as one moves down in group IA?
50.
The position of the elements with atomic number 14 in the periodic table is
51.
Which of the following represents the electronic configuration of the elements in the same period of the Periodic table?
52.
The element is located in a period of .
53.
The energy required to remove an electron is .
54.
Which one of the following is a chemical industry?
55.
Which one of the following is a plant Nutrient?
56.
The direct change of state from gas to solid is called.
57.
In one of the following physical change heat is NOT needed?
58.
Bases change the color of phenolphthalein in to.
59.
Among the following salts which one is a Normal salt?
60.
is obtained by when an a acidic hydrogen is replaced by metal or ammonium ion.
61.
Which one of an acid can produce an acidic salt?